Hi Brandon, What is the best estimate of the ratio of radiation to space taking place from CO2 relative to H2O?
Dunno Rog. MODTRAN could probably get you in the ballpark. This is CO2-only from 70 km looking down, more what I would expect an actual Nimbus retrieval to look like. http://climatemodels.uchicago.edu/modtran/

Although CO2 absorbs thermal radiation from the Earth, it emits more. Carbon dioxide is in thermal deficit in terms of radiative balance. Nitrogen and oxygen constantly feed CO2 with heat so that it maintains a temperature higher than its radiative equilibrium. CO2 is a coolant.
> CO2 is a coolant. Location matters Rog. Again CO2 only, which ignores wv’s significant contribution to backradiation at the surface. A hyper accurate value isn’t required to make the point you can’t simply ignore energy fluxes which don’t fit the pressure-diddit model.

You can keep all your radiative magnitudes, but you’re going to have to give up assuming they’re the cause of atmospheric temperatures rather than the effect. Because CO2 is in radiative deficit unil the temp is down to -77.8°C,
> You can keep all your radiative magnitudes Not if I pull all of the LW emitters out of the atmosphere, Rog. This is where your “CO2 radiates according to its temperature” argument breaks down — not because it’s incorrect, but because of what it leaves out.

You’ve lost me there. What has your computer game got to do with my description of reality?
> You’ve lost me there. Retrace the steps. 1) Sun heats ground. 2) Ground heats atmosphere. 3) LW emitters do so as a function of their temperature. You should only need a crayon and some paper to do the rest of the math here.
Oh ok, I see the problem. You only managed to read the first sentence of my tweet.
> You only managed to read the first sentence of my tweet. Wrong answer, Rog. Even engineers should be able to figure out that 333 less 396 is greater than -396. This really isn’t that difficult.

Ah, I see you’ve been bamboozled by Kevin Trenberth. The radiatively active molecules in the atmosphere radiate according to the temperature they are given by pressurised N2 and O2 in all directions, not just up and down. Here, I’ve fixed the diagram for you:
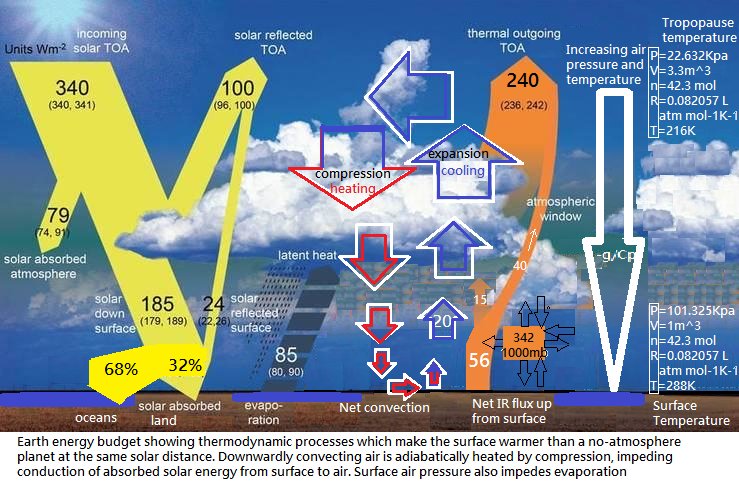
> Ah, I see you’ve been bamboozled by Kevin Trenberth. This looks like 333 W/m2 to me, Rog. Get a new box of crayons and try again.

I can see we’ll need to take this slowly. When you only use up and down meters, you’ll only see up and down readings. Have you heard of something called mean free path length by the way?
> Have you heard of something called mean free path length by the way? You bet, Rog. Have you ever heard of something called “heat-seeking missile”?

Why is the helicopter engine beaming heat only downwards in the illustration? Don’t hot objects radiate in all directions?
Teaching you aerodynamics is quite above my paygrade, Rog. Since that’s ze Moshpit’s brochure perhaps I can let him explain how rotor downwash works. Imma gonna go smoke another bowl, I’m clearly not stoned enough for this.

You’re talking about radiation-thermalised N2 and O2 being forced downwards by air pressure generated by the helicopter blades.. What has this to do with your direction-selective measurements of radiation from the surface being absorbed and re-emitted in all directions by air?
That any computer game used by the USAF to help them detect ICBM launches, and do — you know, AIR FORCE shit — is probably good enough to use for climate science. Between that and your ad hoc fingerpainting it’s a pretty easy choice.

There’s nothing ‘ad hoc’ about the computational proof on the right of the diagram. Check the figures for yourself. Maybe you need to understand the Bernoulli equation a bit better as well as the ideal gas law. http://hyperphysics.phy-astr.gsu.edu/hbase/press.html …

> computational proof I love it when you guys come up with fancier and fancier ways to say “model”. So painted into a corner it isn’t funny … because sadly it seems to be working. Again for the left side of the curve, here’s 333 W/m2 downwelling LW, *observed*. Deal with it.

As I said, no problem with your radiation magnitudes. The problem is you seem to think these measurements tell you something about cause and effect. They don’t. My calcs aren’t a model, they are just quantities which confirm the ideal gas law., and the standard atmosphere.
Brandon R. Gates Retweeted Brandon R. Gates
> cause and effect Which statement on this list is incorrect, Rog.
Brandon R. Gates added,
You missed a step between 2 and 3. CO2 is thermalised in collisions with N2 and O2 and raised from its radiative equilibrium temperature of -77.8C to 15C in the 1 bar pressurised near surface environment. See Kirchoff and Bernoulli, and then report back.

> You missed a step between 2 and 3. I didn’t miss that CO2 is part of the atmosphere, Rog. Once heated by the ground, does it or does it not radiate according to its temperature?
You’re still missing the step. As well as being heated by conduction from the ground, CO2 is being kept warm by collisions with warm N2 and O2 while it is busy trying to radiate it’s way to radiative equilibrium at -77.8C
> collisions with warm N2 and O2 Which was heated by what, Rog. And how does this goose chase lead us to conclude that CO2 suddenly does not emit according to its temperature as you had promised us earlier? How does “mean free path” negate 333 W/m2 of downwelling LW?

You’ve asked three questions, so I’ll answer with three tweets, as I know you only reply to one thing in a tweet. In reverse order: 3) Mean free path means ‘downwelling’ LW doesn’t get directly from cloud base to surface as Trenberth’s misleading cartoon implies. [Tweet 1 of 3]
2) CO2 does radiate according to the temperature it is kept at by warm N2 and O2, despite trying to cool to it’s radiative equilibrium temperature of -77.8C, and this elevated temperature is what we’re measuring CO2 radiating at. [Tweet 2 of 3]
1) N2 and O2 are at the temperature they’re at, because the energy density of the troposphere is what it is, because energy=pressure*volume (and is proportional to insolation), in accordance with Bernoulli’s equation. [Tweet 3 of 3]

doesn’t get directly from cloud base to surface
Oh for shite’s sake, Roger it’s a schematic. Are we supposed to draw every fucking atom in the atmosphere before you’ll rent a clue.
Well at a bare minimum, I’d expect you to acknowledge that the chain of CO2 molecules doing the absorbing and re-emitting are only emitting according to the elevated temperature they are kept at by collisions with warm N2 and O2, in turn kept warm by energy=pressure*volume (+Sun)
via Tallbloke’s Talkshop
October 8, 2018 at 08:27AM


