It turns out that replicating a cow in a laboratory is not as simple as expected. A new study points at some very major and potentially very hard to solve problems with lab meat. We can scale up bacteria in factories easily, but animal cells are very different. Muscle cells not only need a sterile complicated broth but they are basically a sitting-duck feast for any bacteria.
Quote of the day:
“USD 2 billion has already been invested in this technology, but we don’t really know if it will be better for the environment,” Risner said.
Think of a cow as being an entire industrial production campus for meat — to deal with chemical toxins it comes with a customized chemical factory (a liver) and two industrial filter systems (kidneys), and a full immune defense force on a 24 hour watch to deal with the constant flood of microbial contaminants. Cows also have nutrient intake systems to break down grass into separate chemical components which are stored, transported and chemically tweaked to suit. All departments are self repairing, and are equipped with their own laboratory testing, messaging and alert service. The sterile growth conditions of muscle are maintained most of the time in close proximity to dirt and poo. The biological machinery has been road-tested and refined for a half a billion years. Yet somehow we thought we could replicate all that and do it more efficiently in a couple of decades.
Instead of thirty factories, 200 labs, 2000 trucks and sterilized vats of heated pharmaceutical grade goo, we could just use a cow.
It takes a lot of energy to replicate a cow
It’s not enough to kill bacteria in the growth broth, we have to remove the dead body parts of the bacteria too. The outside shell of many bacteria breaks up into is what we call an endotoxin. You may not know it but these are just bad, bad, bad — they are lipopolysaccarides that sometimes leak from our intestinal walls and trigger fever, nausea, inflammation, shivering and shock. So the dead parts of bacteria have to be cleaned out of the broth — which means chromatography, or ultrafiltration, or ion exchanges, and fine membranes. All of which uses lots of energy.
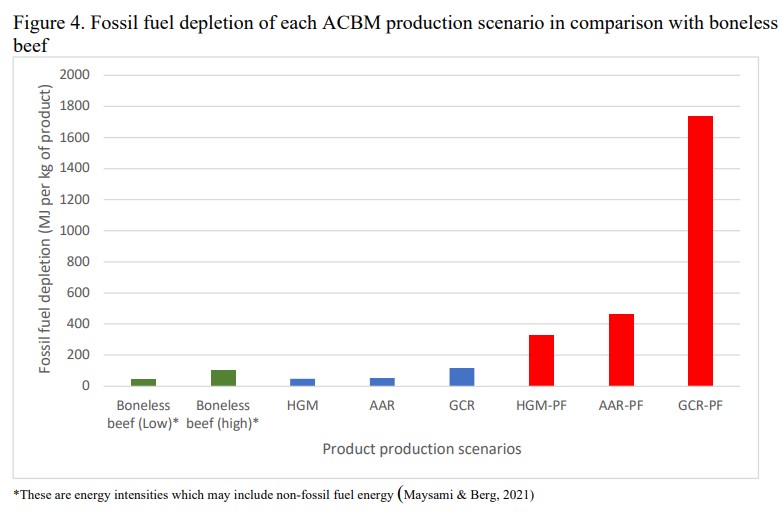
Fake meat practically eats fossil fuels:
The three red bars on the right are different scenarios for creating growth mediums. The PF stands for Purification Factor (meaning highly purified).
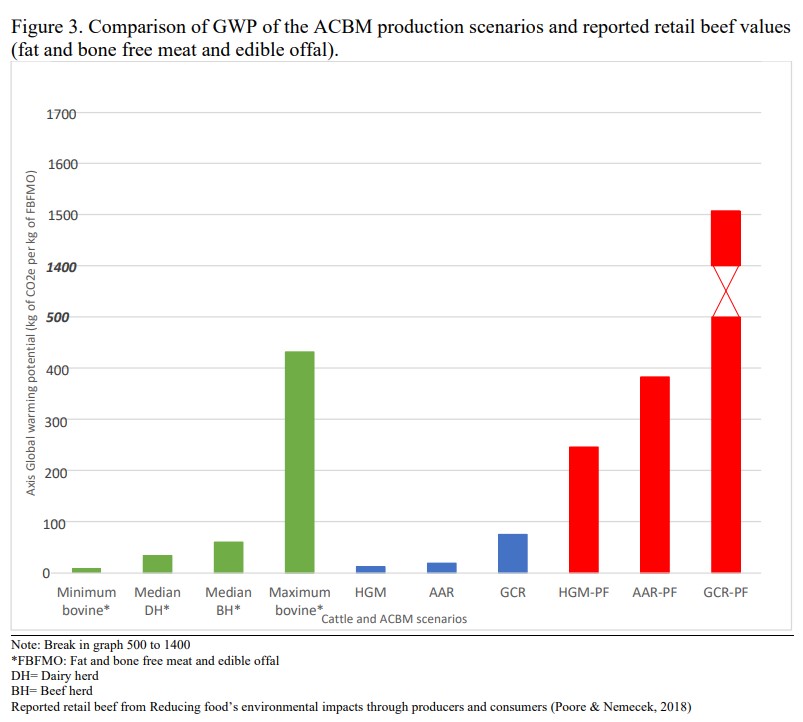
GWP means Global Warming Potential. ACBM means fake meat or “Animal Cell-Based Meat”.BH means Beef Herd, and DH means Dairy. Risner et al
Would you like global warming with your burger?
Note the scale on the vertical axis.
TVP World
The broth itself must contain salts, sugars, amino acids, and vitamins, and the production of each of these elements involves an expenditure of energy.
This “pharmaceutical” level of purification is essential as animal cells will not grow in a broth that is “contaminated” with bacteria. Experts involved in improving the process are testing to what extent it is possible to move away from purification.
The paper’s first author, Derrick Risner of the University of California (U.S.), in a commentary for “New Scientist”, expressed doubt that moving away from “pharmaceutical” levels of broth purification would be possible since even trace levels of contamination can destroy animal cell cultures.
From the paper — we used to use serum from baby cow blood to grow cells in culture but that is an 18 step process and energy intensive…
Animal cell culture is inherently different than culturing bacteria or yeast cells due to their enhanced sensitivity to environmental factors, chemical and microbial contamination. This can be illustrated by the industrial shift to single use bioreactors for monoclonal antibody production to reduce costs associated with contamination (Jacquemart et al., 2016). Animal cell growth mediums have historically utilized fetal bovine serum (FBS) which contains a variety of hormones and growth factors (Jochems et al., 2002). Serum is blood with the cells, platelets and clotting factors removed. Processing of FBS to be utilized for animal cell culture is an 18-step process that is resource intensive due to the level of refinement required for animal cell culture. Thus, the authors believe that commercial production of an ACBM product utilizing FBS or any other animal product to be highly unlikely given this high level of refinement.
The authors say they might be underestimating the costs.
The requirement of endotoxin removal would also contribute to the environmental impact of ACBM products which makes our LCIA results for the minimum scenarios to be underestimated minimums. Utilization of commodity grade growth medium components such as glucose for animal cell growth is unlikely unless the components undergo an endotoxin separation process. The effect of endotoxin can vary greatly depending on cell type and source; however 25 ng/ml of endotoxin was shown to cause cell apoptosis when coupled with non-lethal heat shock (Corning, 2020).
Endotoxins kill cells at just 25 nanograms per ml. The purity required in a complicated mixture on a commerical scale needs Olympic level chemistry. It’s like feeding pharmaceutical grade drugs to your cows instead of grass. This isn’t going to scale up well.
REFERENCE
Risner et al (2023) Environmental impacts of cultured meat: A cradle-to-gate life cycle assessment, bioRXIV, bioRxiv preprint doi: https://ift.tt/67UGFLM;
h/t Another Ian, John Connor II and Climate Depot
10 out of 10 based on 1 rating
via JoNova
May 12, 2023 at 03:30PM